 Often we hear, “The water is what makes it taste so good” when we talk about our favorite foods and drinks. The phrase has been applied to bread, pizza, beer, and virtually any other food. But what about that mug of drip coffee, or perhaps that inky dark shot of espresso? How does the water affect these drinks?
Often we hear, “The water is what makes it taste so good” when we talk about our favorite foods and drinks. The phrase has been applied to bread, pizza, beer, and virtually any other food. But what about that mug of drip coffee, or perhaps that inky dark shot of espresso? How does the water affect these drinks?
To answer this we need to dive into the brewing process. Making coffee involves just a few easy steps. But remember that easy does not mean simple; numerous factors affect your final brew including roast freshness, grind freshness, and water quality.
Water quality is especially important because it facilitates the extraction of soluble components from the coffee grounds into the water; each of the over 600 volatile compounds present in coffee beans are affected by water composition.
In this post, we’ll talk about how brewing water quality affects coffee extraction, methods to measure coffee extraction, and suggest some target numbers for your brew. We’ll focus on the role of total dissolved solids (TDS) in coffee and the brewing water, but remember that there are countless other variables in the brewing process.
What is Total Dissolved Solids (TDS)?
Simply, TDS is the amount of “stuff” (solids) that are dissolved in a given volume of solution. Most commonly, we measure it in parts per million (ppm) or mg/L. Since there are 1000 g in one liter of water, converting to mg would result in 1,000,000 mg per liter of water or ppm.
Notice that TDS measurements are not specific as to what is dissolved, it’s simply the amount of substances dissolved. This means that 100 mg/L of salt and 100 mg/L of sugar would have the exact same TDS value. This has important repercussions for when we discuss TDS measurements later on.
Understanding Dissolved Solids in Coffee
We all know that when we make coffee, we’re extracting some portion of the bean into our brewing water. The portion of the compounds in the bean is what makes coffee the delicious beverage it is. The amount of soluble solids in brewed coffee varies quite a bit, from 1-2% (more on how this varies later). But what exactly is dissolving?
The dissolved solids of coffee include chlorogenic acids, esters, and caffeine, as well as organic acids like citric, malic, and lactic acids. These compounds are what make up a coffee’s body, aroma, and flavor and comprise a coffee’s TDS measurement.
Altering how much of these compounds dissolve into the brewing water gives brewers a wide degree of control over how the finished product turns out. The parameters brewers commonly control include grind size, coffee extraction methods, coffee extraction time, and temperature. However, TDS is an emerging and increasingly more popular metric for controlling coffee strength.
Why does TDS matter?
Since the TDS value correlates to how much stuff is dissolved into a coffee, brewers can use it to estimate how strong the coffee is. More dissolved solids means a more concentrated brew and a more flavorful one. However, there is a limit to how much coffee beans can release into the water before they start releasing less desirable compounds in the coffee, leading to an acrid, over-extracted flavor. Thus, successful extraction starts with quality water.
TDS in your source water
Consider your school science lessons on diffusion in which compounds travel from areas of high concentration to those of low concentrations. Coffee extraction works similarly to this principle with regard to dissolved solids in the brewing water. This means that the amount of total dissolved solids (TDS) in brewing water directly correlates with its extraction efficiency. Water with very low mineral content tends to overextract coffee, while water with high TDS values tends to underextract.
Fortunately, organizations such as the Specialty Coffee Association of America (SCAA) provide guidelines in the form of their brewing water quality standards. The SCAA specifies a TDS level of 75-250 mg/L (ppm) for brewing water, with an ideal target of 150 mg/L. Examples of dissolved solids in brewing water include calcium, magnesium, and chlorides. Brewers can control TDS concentrations in water through water filtration (such as reverse osmosis) and remineralization. These steps help ensure that water is has a consistent TDS value no matter what comes out of the tap.
 Reverse osmosis water filter
Reverse osmosis water filter
TDS in brewed coffee
TDS has important repercussions when developing the ideal ratios for your coffee. Just as the SCAA provides specifications for water, they also have a Golden Cup standard, which provides specifications on making an “ideal” coffee. The specifications are as follows:
Coffee shall exhibit a brew strength measured in Total Dissolved Solids of 11.5 to 13.5 grams per liter, corresponding to 1.15 to 1.35 percent on the SCAA Brewing Control Chart, resulting from a solubles extraction yield of 18 to 22 percent.
Coffee Extraction Yield Calculation
In addition, coffee needs to be brewed in a 55 g/L ratio of coffee to water at 200°F ±5° among other specifications. Note that this is a great starting point for coffee, but you may find that some beans might taste better with even higher or lower extraction percentages. TDS can be measured, but we’ll need to calculate extraction yield.
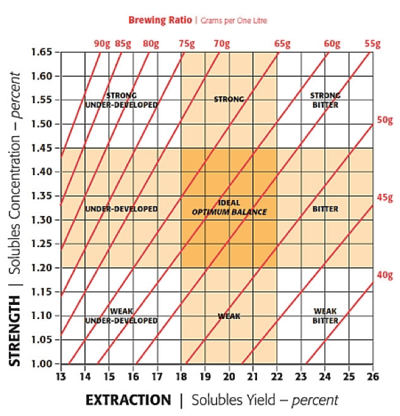 Image: SCAA
Image: SCAA
In order to calculate coffee extraction yield, you’ll need a refractometer (more on this later) and a scale that measures in grams. Afterwards, simply follow this formula:
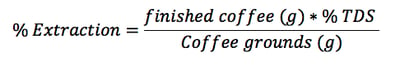
So for example, if you brewed a liter of coffee using the SCAA ratio of 55 g/L and got a TDS of 1.15%, your extraction ratio would be:
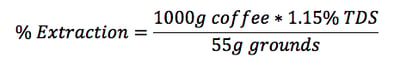
Completing this calculation would yield an extraction of 20.91%, well within our golden cup ratio. If we were a little high or a little low, we could check our temperature, time, grind size, or brewing water. A lot needs to go well for a good extraction!
Implementing Coffee TDS Measurements
Now that you know the impact of TDS on coffee and brewing water, we can go ahead and measure both brewing water and finished coffee for extraction yield. Unfortunately, due to the differences in measurement technology, you must use different meters toaccurately measure the water used for brewing and your finished coffee.
Conductivity meters are used for determining total dissolved solids in water, while refractometers are preferred for finished coffee.
TDS Meters (Conductivity) For Water
Electrical conductivity (EC) meters that also measure TDS excel in measuring low and high levels of ionic dissolved solids (salts), like those in water, but do not detect poorly conductive and nonionic dissolved solids in solution. This includes soluble carbohydrates (sugars), lipids, and other compounds. Since most dissolved solids in clean water only contain minerals and other charged ions, this makes conductivity meters ideal for measuring TDS.
You may notice that we’re talking about conductivity meters for TDS. Nearly all TDS meters measure EC and convert to TDS reading using a conversion factor.
The SCAA specification for water uses a 4-4-2 conversion factor. The 4-4-2 factor is based on the TDS of a 40% sodium sulfate, 40% sodium bicarbonate, and 20% sodium chloride mixture. This factor essentially mimics natural water and is approximately 0.7.
Be sure to look for a TDS meter for your brewing water with the following features:
- Ability to convert using a 0.7 conversion factor
- Measuring range for typical brewing water (0-1000 ppm or more)
- 1 ppm or better resolution
- Waterproof (not essential, but good to have)
Refractometers for Finished Coffee
Refractometers can measure all dissolved solutes by the principle of refraction. When a light passes through from the air into water, it bends (refracts). This happens because the speed of light is different in air than it is in the liquid. When light refracts in this manner there is an angle present where the light no longer passes into the liquid, but rather is reflected entirely off the surface. This angle is called the critical angle. You can see this if you open your eyes while underwater in a pool and look at the surface of the water.
Increased dissolved solids in water increase the refractive index, and as a result the amount that the light bends. Unlike TDS meters that rely on conductivity, both nonionic and ionic soluble particles influence the refractive index. A refractometer measures the critical angle and, using the principles of Snell’s Law, converts this measurement to a solids concentration. Many of these refractometers measure in Brix, a unit common to the beverage industry. However, we can easily convert Brix to % coffee solids by multiplying by 0.85. Researchers at the Bunn Technology Center derived this conversion by comparing with dehydration, the only direct way to measure TDS.
Refractometers can be digital or mechanical. Mechanical refractometers work by placing a sample of coffee on a lens and holding it to a light. Then, you have to look through an eyepiece and judge where the light is no longer visible. The line where that occurs is the result.
 Diagram of a mechanical refractometer's measurement scale.
Diagram of a mechanical refractometer's measurement scale.
Digital refractometers simplify the testing process. They take the guesswork out of judging the result and can be used in low light conditions.
If you are considering a digital refractometer, look for the following features:
- Temperature compensation to help correct for elevated temperatures in coffee.
- Simple operation. Most refractometers only require a few drops of sample and can be calibrated with distilled water in seconds.
- Water resistance to protect from splashes and spills.
 Hanna HI96801 digital refractometer
Hanna HI96801 digital refractometer
Conclusion
TDS measurements are extremely useful indicators of extraction in finished brews, but also have important implications for your source water. Just remember that tasting is essential to any technique refinement – you might find that some beans taste better at higher extractions!
At the very least, it’s important to be sure that the source water measures up to SCAA standards. After all, your coffee is only going to be as good as the ingredients you put into it. We focus on sourcing quality beans, so why not examine the water?
As a leader in innovation Hanna Instruments developed the HALO Wireless pH Meter, which uses Bluetooth Smart Technology to connect to Apple and Android devices running the Hanna Lab App.
Continuing with this tradition, the Hanna Instruments Blog is devoted to sharing the latest in product overviews, how-to guides, and industry specific news to our ever-growing audience.
Contact us at sales@hannainst.com.