Nearly Everything You Need To Know About Electrical Conductivity
Nearly Everything You Need To Know About Electrical Conductivity
Electrical conductivity is a useful measurement in a variety of industries, applications, and situations. Knowing exactly what electrical conductivity (EC/TDS) is, probe options, proper measurement techniques, and how to give probes a bit of TLC when they need it can jump-start your testing.
Navigate the world of electrical conductivity with our Ultimate Guide:
- What is Electrical Conductivity?
- What is Resistivity?
- What Units are Used to Measure Electrical Conductivity and Resistivity?
- Total Dissolved Solids (TDS)
- Salinity
- What Affects Electrical Conductivity?
- Who Tests Electrical Conductivity?
- Types of Electrical Conductivity Probes
- Two Electrode Probes
- Four Ring Probes
- Toroidal Probes
- How To Take an Electrical Conductivity Measurement
- Top Tips for Accurate Electrical Conductivity Measurement
- Tried and True Electrical Conductivity Probe Troubleshooting
What is Electrical Conductivity?
Electrical Conductivity (EC), quite simply, is the ability of a substance or solution to conduct (transmit) an electrical current over a defined area. Electrical conductivity is also known as EC. Current can flow through liquids on an atomic, or ionic level. The ease at which the current is transmitted over a defined area helps to quantify the ionic concentration of a sample. However, the ionic concentration given by an electrical conductivity measurement is non-specific. This means that while you get a measurement of ions in solution, you will not be able to differentiate between different ions.
Ions are charged particles that float free in solution unattached to other molecules. Since ions are charged, they are able to carry and transmit a current. Ions are formed when a solid such as salt is dissolved in a liquid to form electrical components having opposite electrical charges. The sodium chloride separates to form Na+ and Cl- All ions present in the solutions contribute to the current flowing through the sensor and therefore, contribute to the conductivity measurement. Conductivity can be used as a measure of the concentration of ions present in the sample.
Hanna Tip: REMEMBER, electrical conductivity measurements are not specific - they can tell you the concentration of ions, but not necessarily what those ions are.
What is Resistivity?
Resistivity is a substance’s ability to inhibit an electrical current. For low and very low ionic concentration, the measured conductivity becomes difficult and not accurate. Resistivity is more often used to detect trace contamination of things such as ultrapure water. Therefore, the resistivity scale is used to express the results as opposed to fractions. The numbers are exactly the inverse of each other. The reciprocal of 0.10 μS/cm or 1/(0.10 x 10-6 S/cm)] is then 10 x 106 ohms x cm (10 MΩ x cm). This is also commonly referred to as "mega-ohms". Either unit of measurement can be used to state exactly the same value.
What Units are Used to Measure EC and Resistivity?
Electrical resistivity uses the unit of ohm meter or Ω x m. Rather than use the units Ω−1 x m−1, in 1971 the unit “siemens” (symbolized by the capital letter S) was adopted by the General Conference on Weights and Measures as an SI derived unit. The unit for electrical conductivity becomes siemens per meter. The Soemens unit is named after Werner von Siemens, the 19th century German inventor and entrepreneur in the area of electrical engineering. Previously to the siemens per meter unit, mho/cm was used to measure conductivity, where the unit “mho” is a reciprocal ohm. The “mho” is the “ohm” spelled backwards. Because of the history of conductivity, micromho/cm and millimho/cm is commonly translated to microSiemens/cm and milliSiemens/cm because they correspond one-to-one. The unit of measurement commonly used is one millionth of a Siemens per centimeter (micro-Siemens per centimeter or μS/cm). When measuring more concentrated solutions, the units are expressed as milliSiemens/cm (mS/cm). For ease of expression, 1000 μS/cm are equal to 1 mS/cm. Often times conductivity is expressed simply as either micro or milli Siemens.
Hanna Tip: To convert between different units of measure for electrical conductivity is easy.
For Example:
1000 microSiemens/cm (μS/cm) = 1.0 milliSiemens/cm (mS/cm)
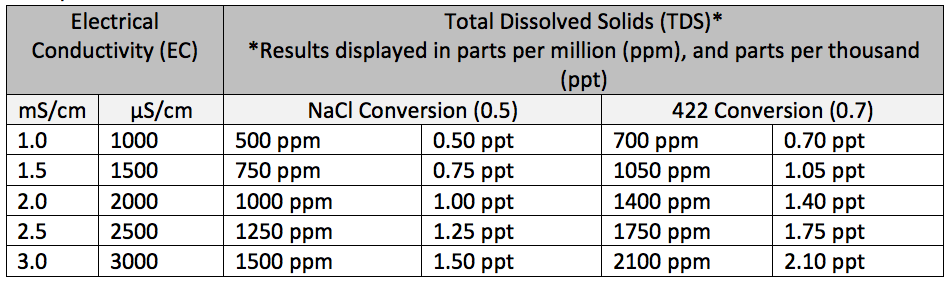
Did You Know Electrical Conductivity Can Be Used to Infer Other Measurements?
While accurate, the following measurements are considered to be indirect measurement methods. This is due to the fact that the meter you use converts the electrical conductivity readings via a conversion factor to obtain the measurements in the desired units.
Example Conversion Table
(Click to Enlarge)
Total Dissolved Solids (TDS)
One EC measurement is called TDS (total dissolved solids). This measures the amount of dissolved matter, both organic and inorganic, in solution. TDS results are in mg/L or g/L. Just remember, electrical conductivity is directly related to the concentration of the ionic dissolved solids. A true TDS measurement is a gravimetric measurement. Every solution has a unique conversion ratio. The conversion ratio is calculated using a known TDS value for the substance then divide that by the measured conductivity value of that same substance. The TDS scale uses 2 μS/cm = 1 ppm (part per million as CaCO3), expressed as 1 mg/L TDS.
Salinity
 Electrical conductivity measurements can be used to determine salinity in different waters. This is very important to remember. It means that an electrical conductivity measurement can only be used to accurately determine the salinity of samples such as seawater. All salinity scales refer back to the salinity of seawater. There are three different salinity scales.
Electrical conductivity measurements can be used to determine salinity in different waters. This is very important to remember. It means that an electrical conductivity measurement can only be used to accurately determine the salinity of samples such as seawater. All salinity scales refer back to the salinity of seawater. There are three different salinity scales.
Salinity Scales
- Practical Salinity Scale (PSU): Established in 1978 by UNESCO and measures salinity on a scale of 0 to 42. This scale is an electrical conductivity ratio and does not have any units associated with it.
- Percent Scale (%): This scale measures salinity on a scale of 0 to 400% with 100% being seawater.
- Natural Salinity Scale: UNESCO defined this scale in 1966, and measures salinity on a scale of 0 to 80 ppt.
Hanna Tip: BOTH TDS and Salinity are considered to be indirect measurements as the meter converts the electrical conductivity reading into the desired units via an algorithm.
What Affects Electrical Conductivity?
The primary influence that can interfere with electrical conductivity measurements is temperature. There are a few select applications where other parameters can greatly influence electrical conductivity (such as measuring soil.)
Temperature
One parameter that can influence your conductivity readings is temperature. This is due to how temperature affects ions in solution. When a solution is at a higher temperature, the ions become excited and the resistance drops and conductance increases. When a solution is cooled, the ions do not have as much energy, so the conductance drops, and resistivity increases. You can also introduce a margin of error if your calibration solutions are not the same temperature as your sample. To compensate for this, probes and/or meters can have a temperature sensor. EC meters can then adjust the readings using an internal calulation to a reference temperature, or Beta. Some meters let you program in your own Beta (temperature coefficient), while others do not. The Beta is different for each substance as ions will behave differently from solution to solution.
| Typical Temperature Coefficients of Various Solutions | |||
| Sample | Percent/°C | Sample | Percent/°C |
| Ultrapure Water | 4.55 | 10% HCl | 1.32 |
| NaCl | 2.12 | 5% H2SO4 | 0.96 |
| 5% NaOH | 1.72 | 98% H2SO4 | 2.84 |
Who Tests Electrical Conductivity?
Everyone can test electrical conductivity, from the home gardener to winemaker to laboratory technician. Here are a few industry examples of why you may want to test electrical conductivity.
Agriculture
Soil (Farming, Greenhouse, Home Gardener)
 If you have healthy soil, you have happy plants. Conductivity is measured to infer the amount of dissolved solids, nutrients, and salinity of soil. Each type of soil has unique EC properties, but it is a good way to tell if you need to fertilize or water your plants more. If conductivity is too low, odds are that you need more fertilizer for your plants, or they have been over-watered. If conductivity is too high, there could be too many nutrients, not enough watering, or salts could have built up in the soil due to the irrigation water.
If you have healthy soil, you have happy plants. Conductivity is measured to infer the amount of dissolved solids, nutrients, and salinity of soil. Each type of soil has unique EC properties, but it is a good way to tell if you need to fertilize or water your plants more. If conductivity is too low, odds are that you need more fertilizer for your plants, or they have been over-watered. If conductivity is too high, there could be too many nutrients, not enough watering, or salts could have built up in the soil due to the irrigation water.
Many things can affect the electrical conductivity of your soil. The most common factors are temperature, soil type and its moisture level, salinity, irrigation and fertilizers, and the depth of the soil. Testing your soil is all about making sure the nutrients are balanced. An EC measurement clues you in to how much is actually there. Remember that EC is good at giving a measurement of the strength of the ions in the soil. This helps you to track the nutrients that are available to your plants.
Plants have varied tolerances to dissolved salts and nutrient concentrations. Plants such as peas and beans are very sensitive to salts deposited in the soil (EC must be below 2 mS/cm). Wheat and tomatoes have a moderate tolerance for higher conductivity. Cotton, spinach, and sugar beets are examples of plants with very high EC tolerances; soil for these plants can go up to 16 mS/cm before damaging crop yield.* It's important to balance the EC of your soil to promote optimal plant health.
*This is referenced from a study that measured EC though a 1:1 and a 1:5 saturated soil extract.
Hydroponics & Fertigation
When growers are looking to fertilize their crops, or to maintain a growth media, it is a fine balance between the nutrients. A great way to monitor these nutrients is to measure EC and TDS regularly. Higher EC and TDS indicate that there are more nutrients available, while lower numbers indicate that nutrient additions should be made. Remember, this number is non-specific – so it won’t tell you which nutrient may be deficient. Fertigators help to have a precise and efficient way to monitor EC and to adjust the nutrients with measured automatic dosing.
Beverage
Beer
 Ground and surface waters contain high minerals and sometimes organic materials and are subject to pollution by human activity. On the other hand, municipal plants are often regulated by law to disinfect or pre-treat water with chemicals, removing contaminants that would adversely affect taste or health. These additives that serve to make drinking water safe can be an issue for brewers as disinfectants can result in off-flavors in the brew. The upside is that TDS can provide you with an indication of the gross mineralization of your water. The downside is that TDS is not ion specific.
Ground and surface waters contain high minerals and sometimes organic materials and are subject to pollution by human activity. On the other hand, municipal plants are often regulated by law to disinfect or pre-treat water with chemicals, removing contaminants that would adversely affect taste or health. These additives that serve to make drinking water safe can be an issue for brewers as disinfectants can result in off-flavors in the brew. The upside is that TDS can provide you with an indication of the gross mineralization of your water. The downside is that TDS is not ion specific.
Monitoring your TDS is a good way to be alerted to any unusual changes in your water supply. Additionally, a high TDS value can indicate water that is more corrosive to equipment and prone to scaling. Water to be used for brewing should have less than 500 ppm TDS.
Some brewers may wish to use selective water treatment options, such as reverse osmosis systems, to remove the minerals from incoming water in order to redesign the water profile. This can be achieved by adding brewing salts and bicarbonates. The ability to define the water profile allows for a diverse array of beer styles.
Wine
If you have ever had a bottle of wine come out of your fridge with a fine floating "dust" swirling around, that wine maker should have done more stringent cold stabilization. The good news is the "dust" is
just potassium tartrate crystals and will not do anything if you consume the wine. Winemakers can cold stabilize their wine through a series of tests, one of them being the monitoring of electrical conductivity. In this test, wine is first held at a set cold temperature, and the first electrical conductivity reading is taken. Then, potassium tartrate (KHT) is added to see if stays unbound (floating around). Then a second electrical conductivity reading is taken. If the electrical conductivity reading changes by 5%, the wine is not cold stable.
Water
Aquaculture
Monitoring conductivity is a good way to check the salinity of an aquaculture operation. For example, appropriate salinity is key to healthy clam aquaculture. Clams are interesting creatures as the salinity of their blood is directly dependent on the salinity of their environment. For them, blood salinity affects their enzyme activity. This enzyme activity directly influences their growth rate and health. For a quick fix, clams open and close their shells to try and control the amount of salinity inside of them. However, if the proper salinity isn’t maintained, you can end up with stunted clam growth or death. 20 to 30 ppt of salinity is considered healthy for clams.
Laboratory
US Pharmacopoeia
USP <645> with Stage 1, 2 and 3 compliance is required for purified water and WFI (water for injection). We offer instruments that are able to perform all three stages required by this standard. Some of these requirements are: Resolution of 0.1 μS/cm or better, accuracy at 1.3 μS/cm of 0.1 μS/cm, to be able to read with or without automatic temperature compensation, the cell constant be known with an uncertainty better than 2%. Conductivity partly depends on other factors such as the pH, the temperature, and the amount of atmospheric carbon dioxide which has been dissolved in the water to form ions (intrinsic conductivity). Conductivity also depends on the chloride, sodium and ammonium ions considered as water impurities (extraneous conductivity). The conductivity (intrinsic and extraneous) of the water is measured and compared to values listed in a table to evaluate if the studied water is suitable or not for use in pharmaceutical applications. If the sample fails Stage 1, additional tests have to be performed (Stages 2 and 3) in order to determine if the excessive conductivity value is due to intrinsic factors or extraneous ions.
Industrial
Plating
While there are many types of plating processes out there, they all have one thing in common: they always will need a rinse bath. Rinse baths help to clean contaminants and residues from the metal pieces in between different points of the plating process. All of the residues introduce contamination to the rinse baths, and eventually, they are too contaminated to use. However, to waste an entire rinse bath takes a lot of time and produces a lot of waste. To minimize this, electrical conductivity is monitored to determine when some additional water may need to be flushed through to keep the baths usable.
Boiler Blowdown
Boilers help heat our homes, businesses, public buildings, and production processes. For large scale boilers, especially those in boiling towers, a lot of feedwater moves through the system. Feedwater has impurities in it, and these impurities can cause issues. As the water is heated it evaporates and becomes steam, and the impurities are left behind. Over time, you will experience a build-up of these impurities, and they can influence the efficiency of the boiler system. Two ways of getting rid of the build up are to test surface water for dissolved solids or treat it, and bottom blowdown where the system is reversed and buildup gets flushed out. Monitoring EC can be used to monitor the TDS and automatically initiate blowdown. This would increase efficiency to the boiler system.
What Types of Electrical Conductivity Probes Are There?
Now that we've gone through what electrical conductivity is, types of tests you can conduct, and why you may be interested in conductivity, let us move on to electrical conductivity probes. There are three types of electrical conductivity probes: two electrode probes, four ring probes, and toroidal probes. You may know them by other names such as: amperometric probes, potentiometric probes, and inductive probes.

Two Electrode Probes
Two electrode probes work through two electrodes that are isolated from each other, but in such a way that they can maintain contact with a sample at the same time. They are made from non-reactive materials such as stainless steel or graphite. The materials need to be non-reactive so the probes do not have side-reactions with the sample, but also so they do not degrade when in contact with a sample (i.e. rust resistance etc.). The two electrodes pass a current at a specific frequency through the sample. Remember what we talked about earlier with ions? That comes into play here. The more ions you have in your sample, the lower the resistance you will have for the current to reach the other electrode. This will result in a higher conductivity reading.
 Two electrode probes are easy to use, relatively inexpensive, require very little sample to get an accurate reading, and you don’t have to worry about the fringe-field effect. Don’t worry, we’ll go over what that is in a bit. With two electrode probes are that the space between the two electrodes has to be stable. If the electrodes get bent, or if a residue builds up, you will get inaccurate readings. Each probe only covers a limited EC range, so if your testing low range and high range samples, you will need to buy multiple probes and/or meters. Also, the higher the EC you go with two electrode probes, the greater the margin of error. The last thing of note on two electrode probes is something called polarization effect. The polarization effect happens when a charge builds up between the two electrodes. This extra charge can cause your EC readings to be lower than they are expected to be. This can be minimized by two electrode probes that have their electrodes made out of graphite instead of stainless steel.
Two electrode probes are easy to use, relatively inexpensive, require very little sample to get an accurate reading, and you don’t have to worry about the fringe-field effect. Don’t worry, we’ll go over what that is in a bit. With two electrode probes are that the space between the two electrodes has to be stable. If the electrodes get bent, or if a residue builds up, you will get inaccurate readings. Each probe only covers a limited EC range, so if your testing low range and high range samples, you will need to buy multiple probes and/or meters. Also, the higher the EC you go with two electrode probes, the greater the margin of error. The last thing of note on two electrode probes is something called polarization effect. The polarization effect happens when a charge builds up between the two electrodes. This extra charge can cause your EC readings to be lower than they are expected to be. This can be minimized by two electrode probes that have their electrodes made out of graphite instead of stainless steel.
Four Ring Probes
A four-ring probe works a bit differently than the two electrode probe. Instead of having two electrodes that pass an alternating current back and forth, this probe has four platinum rings on the body of the electrode, as well as a vent hole. The top and bottom rings act as drive electrodes for the probe. These two rings apply an alternating voltage the sample, and this induces a current. The two center rings are your sensor rings, or electrodes. They measure the potential drop in the current generated by the drive electrodes. A potential drop is the difference in voltage between two points in a circuit. As a sample is introduced to a sample, the potential drop changes in proportion to the conductivity.
When using a four ring probe, you only need one probe to cover your entire testing range, up to 1 Siemen per centimeter, and they are more accurate at high levels of conductivity than the two electrode probes. You also do not have to worry about the polarization effect. The construction of this probe, with how the rings are situated, causes a constant field of current to be maintained around the rings; it acts as a shield. For the four ring probe you will have to use a larger sample volume to adequately submerge the probe, and it is a more of a financial investment. While you don’t have to worry about the polarization effect, you do need to be wary of the fringe field effect. This happens when the measurement field, that constant current, extends outside of the probe. You only need to worry about the fringe field effect if your probe is too close to the sides of the container or pipe where you are taking an EC measurement. A good rule of thumb is to keep the probe at least an inch away from all surfaces. The distance you need to keep the probe varies, so just check the manual.
Toroidal Probes
Toroidal probes are sometimes called non-contact probes, or electrodeless probes. The require a control processor. Therefore, they are used in process equipment. The probe looks a bit like a doughnut, as it is constructed of two inductively coupled coils stacked on top of each other. The two coils are encased in a chemically resistant plastic sheath. One coil acts as the driver electrode that applies a current (a magnetic field), while the second is the receiver or sensor electrode. Then, changes in the field are monitored.
Toroidal probes are great because the sample never actually touches the sample, since they’re encased in plastic. You don’t have to worry about the polarization effect or the fringe field effect. The probe has a very low chance of fouling or getting clogged, and you do not need calibration solutions. They are very accurate in high range samples, up to 2 Siemens per centimeter. The plastic body is chemically resistant. However, toroidal conductivity probes are expensive, and not suitable for low-range EC samples. Even though the plastic sheath is chemically resistant, it is not impervious. Be careful not to have organic solvents in your sample, or concentrated chlorine. Select plating baths can also cause issues due to incompatibility with the plastic.
| Electrical Conductivity Probes: A Comparison | ||
| Probe Type | Pros | Cons |
| Two Electrode Probes | Inexpensive,. Small sample volume. No fringe field effect. | You need a different meter/probe for each testing range. Polarization effect. |
| Four Ring Probes | One probe covers your whole testing range. No polarization effect. | Fringe field effect. Larger sample volume is needed. More of a financial investment. |
| Toroidal Probes | Higher testing range. Chemically resistant. | Lower accuracy in lower range samples. Most expensive out of the three types of probes. |
How To Take An Electrical Conductivity Calibration or Measurement:
When calibrating, we recommend following the subsequent steps:
You will need: two beakers, a calibration standard, meter, EC probe, and deionized water.
- Before even placing a probe into a standard.
a. Rinse the probe in deionized water (distilled water can work, but do not use tap or drinking water).
b. Gently shake off the excess water from the probe.
c. Immerse the probe in a beaker or cup with some standard.
d. Swirl the probe and then remove it from the beaker.
e. Repeat steps c and d 3-5 times to “clean” the probe. - Next, you can calibrate the probe in a standard.
a. Pour a fresh measure of standard.
b. Turn on the meter.
c. Immerse the probe in a beaker or cup with some standard.
d. Swirl the probe and then remove it from the beaker.
e. Repeat steps b and c 3-5 times to “clean” the probe.
f. Swirl and tap (gently) the probe on the bottom of the beaker. This removes any trapped air bubbles.
g. Gently stir the solution, you don’t want to introduce extra air bubbles into the standard.
h. Wait for the probe to reach thermal equilibrium (wait for the temperature to stabilize).
Measuring a sample follows the same steps for two electrode probes and four ring probes. Just substitute your sample for the calibration standard and you’re good to go.
10 Top Tips for Accurate Electrical Conductivity Measurement
Here at Hanna, we have had years of experience with electrical conductivity testing. We have found that there are ten top points to remember when testing.
 Tip #1: Proper probe submersion.
Tip #1: Proper probe submersion.
Incomplete probe submersion can cause erratic, as well as inaccurate electrical conductivity readings. The amount of probe that you need submerged varies between types of electrical conductivity probes. Two electrode probes require a smaller sample volume where only the sensing portion of the probe needs to be covered. Four electrode probes require a larger sample volume where the vent hole above the rings must be submerged.
 Tip #2: Use a plastic beaker.
Tip #2: Use a plastic beaker.
This tip is particular to using a four ring probe. Remember how we mentioned the fringe field effect earlier? To help minimize the effect even further, we recommend using a plastic beaker when you are performing a calibration or measurement. Glass or metal containers can cause EMC interferences (also known as electromagentic interferences). Use a plastic beaker, and keep the probe at least one inch away from all sides of the beaker.
Tip #3 Always rinse your probe.
Salt build-up and other residues can contaminate your calibration buffers and samples. This will cause inaccurate and erratic readings. Follow the directions for rinsing your electrical conductivity probe found above in How To Take An Electrical Conductivity Calibration or Measurement.
Tip #4: Always properly store your probe.
Storing a regular electrical conductivity probe is easy. Ensure the probe is clean, and then store the probe dry. However, if you have a combination (electrical conductivity/pH) probe, you must follow certain storage steps to preserve the integrity of the probe. If a combination probe is stored incorrectly, the pH glass can dry out, and this will cause inaccurate readings. When using a combination probe, always store the probe in pH storage solution. You can always reference the manual to determine the exact type.
Did you accidentally store a combination probe dry? No worries! You can revive the dried out pH glass by soaking the probe in pH storage solution for a minimum of one hour.
Tip #5: Clean your probe regularly.
Just like with a pH probe, electrical conductivity probes require specific steps to keep them clean and in tip-top shape. Deposits, salts, and other contamination can accumulate on your electrical conductivity probe over time. Accumulation can interfere with the electrode portion of the probes, as well as the vent hole on four ring probes. This can cause reading issues as well as calibration errors. Use warm water with some soapy surfactants to remove deposits. Then, rinse the probe thoroughly with deionized water. Do not use cleaning solutions or solvents as they can damage the probe.
If you have a combination probe, where electrical conductivity measurements and pH measurements can be taken with the same probe, please reference the pH guide.
Tip #6: Calibrate often.
Properly calibrating your electrical conductivity probe helps you to achieve accurate results. Electrical conductivity probes can either be calibrated to a single calibration point, or to multiple calibration points. If the probe is used every day, then the probe should be calibrated daily. If the probe is used less frequently, then calibrate the probe prior to use.
If you have questions about calibration, check out the How To Take An Electrical Conductivity Calibration or Measurement section above or take a look in your user manual for your particular probe.
Tip #7: Always use fresh calibration standard.
Electrical conductivity calibration standards have no buffering capacity. This means that the standards are easily contaminated. Any contamination introduced to the calibration standards will cause errors during the calibration process. To avoid contamination of the calibration standard, always use fresh standards. For specific calibration instructions, please reference the How To Take An Electrical Conductivity Calibration or Measurement section above.
Tip #8: Check for bubbles.
When submerging a four ring probe, you sometimes may get air trapped between the internal probe, or you may have air bubbles stay on the surface of the probe. and the probe sleeve. Bubbles may seem like not a big deal, but they can cause erroneous calibrations and readings. To get rid of bubbles, submerge the four ring probe past the vent hole. Then, move the probe up and down in the sample or calibration standard. You can also gently tap the probe on the bottom of the beaker to dislodge any bubbles that have not yet dissipated.
Tip #9: Pick the right probe for your sample.
Keep in mind that not all electrical conductivity probes can measure across all measurement ranges. For example., two electrode probes only cover the specific range they were designed to measure. Get to know your sample. If your sample range varies, then you will need a probe that covers the entire possible range.
 Tip #10: Take the time for stability.
Tip #10: Take the time for stability.
No matter how accurate a probe or meter is, it still will take an extra moment or two for the readings to stabilized. This is true even if your probe has an integrated temperature sensor. Taking the extra time will pay off and help to ensure you get true readings.
What Should You Do If You Run Into a Problem?
Tried and true electrical conductivity probe troubleshooting techniques are always good to have in your toolkit. Below are the top 3 problems that people run into when testing electrical conductivity. Each problem will be answered with solutions to guide you through how to troubleshoot your electrical conductivity probe and meter.
Problem #1: My readings keep drifting or are erratic.
Question: Is your probe dirty?
Depending on your sample, debris could be liable to stick to the probe. Clean the probe regularly to prevent buildup between the electrodes (two electrode probe) or blockage of the vent hole (four ring probes). Rinse the probe in between readings, and before/after storage in order to help prevent buildup. Especially when working with water, residue may not be visible to the naked eye, so when in doubt, rinse.
Question: Is the probe set up correctly?
Make sure that the probe connector is properly connected to the meter (if using a probe with a cable). For 3.5 mm connectors, ensure that the pin is inserted all the way into the port. For DIN/Quick DIN connectors, double check that the pins are lined up properly.
 If your probe is a new four ring probe, take a look inside the probe sleeve near the bottom. There is a small clear rubber circle around the internal section of the probe. This is to help preserve the probe during shipping, but it can interfere with your readings. Carefully remove the rubber circle, then re-calibrate the probe.
If your probe is a new four ring probe, take a look inside the probe sleeve near the bottom. There is a small clear rubber circle around the internal section of the probe. This is to help preserve the probe during shipping, but it can interfere with your readings. Carefully remove the rubber circle, then re-calibrate the probe.
Question: Did you choose the right probe for your sample?
Check that your probe is correctly submerged. Two electrode probes only need to be slightly submerged to cover the plates. Four ring probes require that the vent hole above the rings be submerged as well.
Make sure that there is at least one inch of clearance between the probe and all sides of the beaker.
Check the usable measurement range for your two electrode probe. If your sample is out of range you need a four ring probe.
Question: Did you calibrate?
Proper and frequent calibration is key. If the probe is used daily, calibrate daily. If not, calibrate the probe prior to use.
Problem #2: My readings are inaccurate.
Question: Did you remember about the Polarization Effect?
The Polarization Effect happens when a charge builds up between the two electrodes. This extra charge can cause your EC readings to be lower than they are expected to be. This can be minimized by two electrode probes that are made out of graphite instead of stainless steel.
Question: Did you remember about the Fringe Field Effect?
The Fringe Field Effect s when the measurement field, the constant electrical current, extends outside of the probe. You only need to worry about the fringe field effect if your probe is too close to the sides of the container or pipe where you are taking an EC measurement. A good rule of thumb is to keep the probe at least an inch away from all surfaces. The distance you need to keep the probe varies, so just check the manual.
Question: Did you calibrate?
If you are testing for USP standards, or in aggressive chemicals, you will need to calibrate more often. If the probe is used daily, calibrate daily. If not, calibrate the probe prior to use.
Question: Did you calibrate correctly?
Check out our Maintenance Guide to verify that you followed the proper calibration steps.
Question: Did you use the right calibration standard?
When doing a single point calibration, calibrate in the air first (zero point) and then the calibration standard used should be as close to the actual concentration of your sample as possible. If your sample concentration fluctuates regularly in a wide range, you may need a probe and meter that can calibrate to multiple standards to increase your accuracy.
| Code | Electrical Conductivity Value @ 25°C | Code | Electrical Conductivity Value @ 25°C |
| HI7033L | 84μS/cm | HI7030L | 12880 μS/cm |
| HI7031L | 1413 μS/cm | HI7034L | 80000 μS/cm |
| HI7039L | 5000 μS/cm | HI7035L | 111800 μS/cm |
Question: Did you reach Thermal Equilibrium?
Even with temperature compensation, it takes time for the probe to reach a stabilized temperature state. Make sure to allow a few minutes for the probe to reach this equilibrium.
Question: Did you get rid of air bubbles?
Air bubbles can wreak havoc on readings as the sensing parts of the probes may not be completely submerged in the sample. Gently swirl the probe and/or tap the probe on the bottom of the beaker. This should be enough to dislodge any trapped bubbles.
Question Maybe what's in your analyte is non-ionic?
Not all things that cause TDS break apart into ions while in solution. Double check that your analyte breaks apart into ions when in solution. If it does not become ionic, there could be other means of testing available.
Problem #3: My probe will not calibrate.
Question: Are you using a contaminated standard?
Remember that the calibration standards have no buffering capacity. Try taking some new/clean beakers and pouring fresh standard into each. Then, use one beaker as an additional “rinse” to help keep the standard for calibrating as free of contamination as possible.

Question: Is your rinse water contaminated?
EC probes are very sensitive, and as mentioned above, the calibration standards are easily contaminated. Old rinse water, or water from the tap, can introduce contaminants. Distilled water can be used, with deionized water being the best option.
Question: Did you store your probe properly?
EC probes should be stored dry unless they are a combination EC/pH probe. If you have a combination probe, the probe should be stored in HI70300 storage solution in order to keep the pH glass probe hydrated and in working order.
Remember to: Clean Regularly, Calibrate Often, Care Always
Have questions?
Contact a Hanna Technical Specialist at sales@hannainst.com, the chat in the lower right-hand corner of the screen, or contact your local Hanna office.
That's why we've dedicated our blog as a helpful resource for you to use! Catch up on the latest products, explore industry trends, discover testing tips, learn how to improve results, and more. Got questions? Email sales@hannainst.com.