
We measure pH for a variety of reasons. From ensuring the chlorine in pool water is an effective sanitizer, to making sure fermented foods are safe to eat, and many other reasons, a well-maintained pH level is critical to many things we do and use daily.
Because pH is so important, we need to make sure we are accurate in our testing. While common pH tests like chemical test kits and pH strips are simple and inexpensive, they come with some issues that could lead to inaccurate results.
Why do we measure pH?
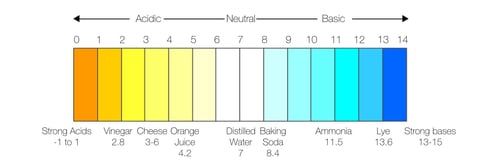
pH stands for the "potential of Hydrogen" and is the measurement of how acidic or basic (alkaline) a substance is. The reading is taken based off of the ion activity in aqueous samples (things containing water). The pH scale ranges from zero to 14, with zero being the most acidic and 14 the most basic; pure water is completely neutral, with a pH of 7.
Measuring pH is done for many different reasons, and your desired pH range is going to depend on what you want to accomplish. Reef aquariums should be at a range of pH 8.0 - 8.3 to keep fish and corals healthy; you want beer mash to be around pH 5.2 - 5.6 to prevent bacterial growth and keep the yeasts healthy; and a desired soil pH range is going to vary based on climate, types of plants, and a few other factors.
In certain cases, a reading of a general pH range might be okay, but for most applications you want your tests to fall into a fairly small window. There are many methods for testing pH, but which one will work best for you?
Testing pH with color matching methods

The first thing you might think of when you hear “pH testing” is the paper strips from your high school chemistry class, or chemical kits with reagent drops used to test pool water. Both of these methods of testing pH give you results based on a chemical reaction that results in a color change. When your paper or liquid sample changes color, you match it to the color guide provided and get your pH reading. These tests are all easy and inexpensive to perform, but they can cost you in accuracy.
As we said earlier, many optimal pH ranges fall within a pretty narrow window, and these types of tests give a broader range -- usually from .25 to one full pH point! Other things like how old the kit is, as well as its exposure to air, sunlight, and extreme temperatures, will affect the accuracy of the reading.
The biggest problem with accuracy in these kinds of pH tests, however, is the subjectivity that comes along with color matching, and how humans perceive color in different ways.
Color subjectivity

The way color is perceived from person to person, as well as in different environments, will vary for a number of reasons. (Just look what happened with that viral dress photo a few years ago.)
Color isn’t as objective as most people think. We see things as certain colors because of the way the object we are looking at absorbs and reflects light. An apple is red because it reflects the red wavelength of light and absorbs all the others; black objects absorb all light, while white objects reflect all light. When different people see an object, their brain can process these light reflections differently than the next person.
Outside elements will also change the way a color is seen, another factor that can interfere with using a color matching pH test. The amount and type of light in the environment as well as other colors close by, and even fatigue all can affect how you perceive color.
Because of these reasons, color matching pH tests can have huge variations in results depending on who is doing the matching, what kind of room you are standing in, and even the time of day.
How good are you at color matching? Take this quiz to find out!
Alternatives to pH color matching
In order to get the most accurate pH results a digital testing method like a pH pocket tester or a pH meter and electrode are the best way to go. There are many different types of digital pH meters on the market that will fit into any application or budget.
Pocket Testers (Perfect for Pools!)
A pocket pH tester is a simple, all-in-one instrument for pH testing. Testers can range from a less expensive general use meter to a more specific version for more specialized uses.
If you’re testing samples that are not abrasive and have a relatively mild temperature, a simple one-button pH tester will work great for you. Automatic calibration and the replaceable electrode make this type of tester easy to maintain.
If you’re worried about dropping your tester in large samples, like a pool, a waterproof tester will give you some peace of mind. While the waterproof testers don’t come with replaceable electrodes, they do feature renewable cloth junctions that allow you to refresh the electrode from time to time and keep it working like new.
Portable pH Meters

Portable pH meters are more expensive than pocket testers for a few reasons. The meters themselves are much larger, and unlike testers the electrode is connected to the meter by a cable or wire. Many also come with more features than a tester, depending on which one you get like Cal Check, a specialized electrode, or the ability to test additional parameters in addition to pH.
Portable meters are generally more specialized than a pocket tester because they come equipped with electrodes and settings that are specific to what you are testing. A portable meter is a step up from testers who want to get more specific with their testing.
Wireless pH Meters

Wireless pH meters like HALO are a good choice for those who want to upgrade from their tester, but are not yet ready for a portable meter. With HALO, you already have a pH meter in your pocket with the Hanna Lab App for iOS or Android. (Download it now to try out HALO in demo mode!) All you need to do is decide which of the ten HALO models is right for you, and connect it to your phone or tablet via Bluetooth.
Want to learn more about digitally testing pH?
The team at Hanna has seen a lot of good (and bad) pH measurement practices throughout our 39 years in business, so we’ve compiled a list of the top 10 mistakes in pH measurement, as well as advice on what to do instead.
That's why we've dedicated our blog as a helpful resource for you to use! Catch up on the latest products, explore industry trends, discover testing tips, learn how to improve results, and more. Got questions? Email sales@hannainst.com.


