
Importance of pH Overall
Just like the lead in a show, pH plays an active role in every stage of the beer brewing process. There are many ways to test pH on the market - everything from test strips to meters! Let us guide you through when to test pH during brewing, and a few recommendations to help you get started! For more particular information on pH probes, maintenance, and more check out our pH guides.
Why pH Matters in Brewing
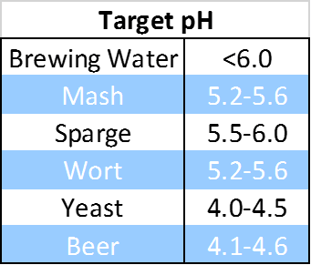
pH, the measure of hydrogen ion concentration in a solution, is essentially a measure of whether the substance you're testing is an acid or a base. Reading pH is important at every stage of the brewing process, from water, to mash, to wort, to yeast, to fermenting beer and finished beer. High pH can lead to harsh off flavors, and poor enzymatic conversion of the mash. Normal beer pH [4.1-4.6] inhibits certain spoilage organisms, while much lower pH may be indicative of infection by acid producing bacteria, resulting in sour beer.
Consistency in the Process
To achieve consistent beer, brewers should know the pH at every step of the process. Water is treated for general potability and is usually out of the necessary range for brewing. Targeting the correct pH range for brewing water and the mash is the first step to brewing consistency, and will ensure that the following steps stay within specifications.
Checking Source Water
Checking on the Water Your Town Supplies
Water supplied by most municipalities is basic and needs to be adjusted to the proper pH range for brewing. Brewers should ask for, or get water analysis online from the municipality that supplies their brewing water. If the water profiles are hard to come by, or if they change frequently, you can also run some tests in house such as hardness, alkalinity, and acidity. This is the starting point for the brewing process and will tell the brewer how much adjustment to the source water is needed . Brewers may also may want to make further adjustments to replicate source water of famous brewing centers like Burton in the UK, Dortmund in Germany and Pilsen in the Czech Republic.
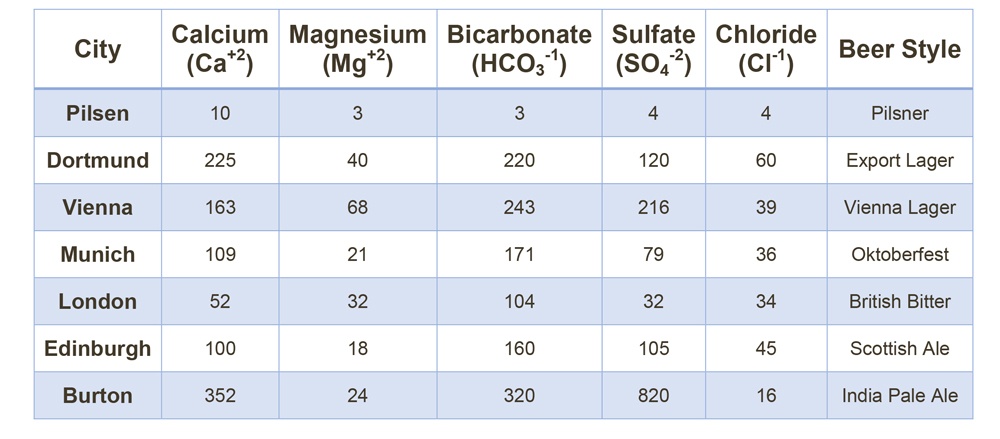 (Table 1)Palmer, John J. How to Brew: Everything You Need to Know to Brew Great Beer Every Time. Brewers Publications, a Division of the Brewers Association, 2017.
(Table 1)Palmer, John J. How to Brew: Everything You Need to Know to Brew Great Beer Every Time. Brewers Publications, a Division of the Brewers Association, 2017.
Multiple online sources have conversion tables to tell you how much calcium, magnesium sulfate, or chloride needs to be added to the water to match various historical water profiles.
pH Adjustment for the Optimum Water Profile
Malt is acidic and will lower mash pH, but the volume of water needed to sparge, or rinse, the fermentable extract out of the grain is significant. Adding food grade phosphoric acid, lactic acid or calcium sulfate can help correct the pH of the sparge water. pH measurements should be taken of brewing water, mash and sparge water to ensure the mash and wort are in range.
pH During Mashing
Enzymes Need the Right Temperature and pH
 Milled malted barley and other grains are mixed into a Mash Tun with hot water [145-156° F] in a process called mashing. Mashing results in proteolytic and amylolytic enzymatic activity which produces amino acids and maltose [fermentable extract] respectively, compounds that will be utilized by yeast later in the fermentation stage. The enzymes are very particular when it comes to pH. There are many resources available on the web that give pH recommendations depending on the beer that you are brewing.
Milled malted barley and other grains are mixed into a Mash Tun with hot water [145-156° F] in a process called mashing. Mashing results in proteolytic and amylolytic enzymatic activity which produces amino acids and maltose [fermentable extract] respectively, compounds that will be utilized by yeast later in the fermentation stage. The enzymes are very particular when it comes to pH. There are many resources available on the web that give pH recommendations depending on the beer that you are brewing.
Wort Stabilization Stabilization Starts in the Mash Tun
Mash in water temperature and mash pH are of critical importance for proper conversion of starch and protein during the mash. Setting the pH specification at the first step of brewing ensures that subsequent brewing and fermentation proceed in the right direction.
pH During the Kettle Boil
How Hop Utilization and Hot Break are Affected
Once the fermentable extract from the mash has been runoff to the kettle, it is called wort. If the wort is in the acceptable pH range, other important changes occur. Hops are added for bitterness and aroma, and utilization of the bittering substances [α-acids] takes place at the elevated temperature of the boil. Heat and wort pH will affect the isomerization of the α-acids to iso- α-acids with a slight advantage of higher utilization at higher wort pH [5.8].1
Hot break, or the coagulated protein from the wort will floc out after the boil, making it easier to produce clean, clear beer after fermentation. While a certain amount of amino acids are necessary for yeast growth and cell membrane production, the majority of protein left behind from the boil could be problematic if left in suspension, causing haze, binding yeast, and eventually precipitating out in finished beer.
Fermentation pH
Fermentation is the Most Critical Phase of Brewing
This is the stage of the process where wort is vulnerable to spoilers, and wild yeast strains. Sterile, oxygenated wort needs to be at the correct temperature and pH range for brewer’s yeast to act on the nutrients created during the brew. pH that is lower than expected during fermentation [pH<3.6-3.7] may indicate serious contamination problems.
Yeast Viability and Performance
Once yeast is added to cooled, oxygenated wort, fermentation begins. Yeast will reduce malt sugars in the wort, giving off CO₂ and alcohol. Yeast will grow exponentially in the presence of wort and oxygen utilizing most of the sugars in the process. The pH will also drop as various organic acids are produced by the yeast.2 How well the yeast performs is related to the parameters mentioned, along with general viability. Checking the pH of yeast before repitching it into the wort is recommended. A tired yeast slurry will have a higher than normal pH [>4.5] due to cytoplasm released by autolysed yeast. Healthy yeast will respond positively to clean, cold, oxygenated wort with adequate nutrients and no competition.
Finished Beer pH
The Right pH in Finished Beer for Shelf Life and Flavor Profile
pH at the final stage of production is an indication of stability. Finished beer should have a lower pH than pre fermentation wort. Any variance of the final pH, especially a much lower reading, may indicate infection that could lead to sour, hazy beer. If the reading is too high at this stage, something from early stages of the process was missed or there is something alkaline that got introduced into the beer.
Brewery Wastewater Issues
High or Low pH and Municipal Wastewater Standards
The acidic nature of beer and the residual caustic quality of cleaning solutions can prove to be a shock to municipal water treatment facilities. Many water treatment plants count on uniform effluent with no spikes of acidity or causticity in the wastewater stream. For example, Framingham, MA Municipal Wastewater Regulations prohibit discharge with pH lower than 5.5 and higher than 12.0, allowing deviations from these limits for facilities that continuously monitor pH.3
Brewery Effluent Can (and Should) be Treated
Smaller breweries may need to hold effluent in a tank and adjust pH before releasing it to the sewer. Monitoring the pH usually means keeping records for the municipal authority. Settling tanks may also be required. Larger breweries sometimes have to install significant water treatment equipment, essentially replicating a municipal wastewater facility.
What to Test pH With
As mentioned above, pH can be tested a number of ways. By and large, the most efficient way to test the pH of beer, and to avoid interference, is with a pH electrode. pH electrodes are comprised of a meter, an electrode body, and pH sensitive glass. An advantage to using a pH electrode is that they can be tailored to your specific application (brewing beer). This specificity allows for things like anti-clogging junctions,, and titanium bodies. The HALO® Wireless Beer pH Meter is an example of a completely portable electrode that is tailored to testing in beer. It turns any Apple or Android device into a laboratory grade pH meter! Another example is the HI981031 Beer pH Tester., designed to be simple and durable for beer testing.
When in doubt, test your pH!
Let us help you in your testing needs. For assistance in choosing the best pH option, please contact us using one of the channels below.
1.IMPROVING THE HOP UTILIZATION IN THE BEER BIOTECHNOLOGY, Bulletin UASVM, Agriculture 65(2)/2008].
2FACTORS RESPONSIBLE FOR THE DECREASE IN pH DURING BEER FERMENTATIONS, J. Brew., May-June, 1976, Vol. 82, pp. 149-153
3Town of Framingham Department of Public Works Wastewater Regulations, May 2015
That's why we've dedicated our blog as a helpful resource for you to use! Catch up on the latest products, explore industry trends, discover testing tips, learn how to improve results, and more. Got questions? Email sales@hannainst.com.




